|
This results in a more intricate pattern of lines in the emission spectra of molecules compared to the simpler spectra of individual elements. The emission spectra of elements and molecules are different because the energy levels in molecules are more complex due to the additional vibrational and rotational energy levels. When a molecule undergoes a transition between energy levels, it can involve changes in electronic, vibrational, and/or rotational states, leading to a more complex emission spectrum. Helium and at least one of the following: Mercury, Neon, water vapor, and Nitrogen. This is because molecules have additional energy levels associated with their vibrational and rotational motions, in addition to the electronic energy levels. electronic energy levels by measuring the atomic emission spectra of. The difference between the emission spectra of these molecules and those of the elements observed previously is that the molecular spectra consist of bands of lines rather than discrete lines. Build and calibrate a simple spectroscope capable of measuring wavelengths of visible light. Some of the most prominent wavelengths are approximately 2.7 µm and 4.3 µm. Carbon dioxide: The emission spectrum of carbon dioxide consists of several bands of lines, with dominant colors in the infrared region. Some of the most prominent wavelengths are approximately 391 nm (violet-blue) and 428 nm (blue).Ģ. Molecular nitrogen: The emission spectrum of molecular nitrogen consists of several bands of lines, with dominant colors in the blue and green regions. Instead of getting a continuous spectrum, you get a discontinuous spectrum composed of a series of colored lines. Unlike blackbody radiation, the light in these tubes is not a continuous spectrum. Now, let's describe the atomic line spectra for the molecules:ġ. neon, or argon, or even sodium through a prism. Pure hot gases can also give off light, like those found in neon tubes. Since each element has a unique set of energy levels, the wavelengths of the emitted photons (and thus the emission spectra) are different for each element. When an electron transitions from a higher energy level to a lower one, it emits a photon with a specific wavelength corresponding to the energy difference between the two levels. The energy levels of atomic orbitals are quantized, meaning that electrons can only occupy specific energy levels. The emission spectra of these elements (and for hydrogen and helium) are different because each element has a unique electronic configuration. 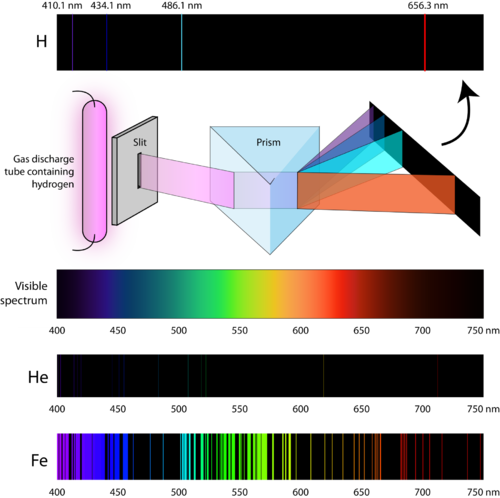
Some of the most prominent wavelengths are approximately 436 nm (blue), 546 nm (green), and 577 nm (yellow). Mercury: The emission spectrum of mercury consists of several bright lines, with dominant colors in the blue, green, and yellow regions. These lines have wavelengths of approximately 589 nm and 589.6 nm.ģ. Sodium: The emission spectrum of sodium is dominated by two very bright lines in the yellow region, known as the sodium D lines. Some of the most prominent wavelengths are approximately 585 nm (yellow), 640 nm (red), and 703 nm (deep red).Ģ. Neon: The emission spectrum of neon consists of several bright lines, with dominant colors in the red and orange regions. 
(b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project.First, let's describe the atomic line spectra for each element:ġ. \): The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
